 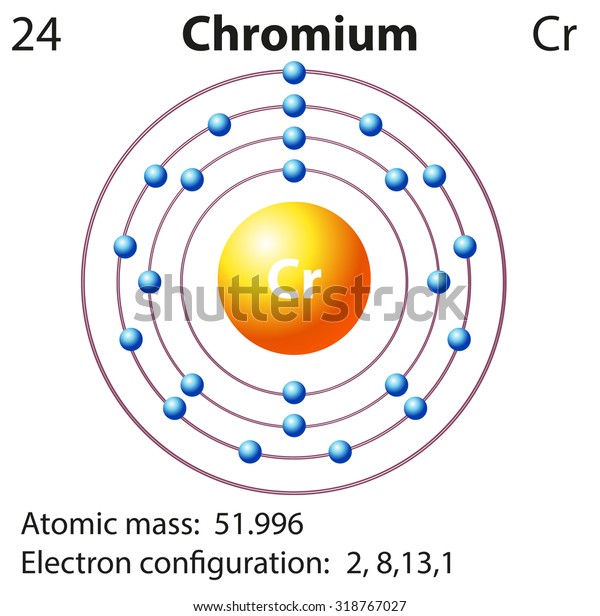
Ask your instructor which method he or she prefers. Using this method, the electron configuration for chromium would be written as #"4s"^1"3d"^5#.Įither way is acceptable, however placing the 3d sublevel before the 4s sublevel is more common. NOTE: Chromium is an exception to the rules for writing electron configurations Video: Cr, Cr2+, and Cr3+ Electron Configuration Notation Electron Configuration for Cr, Cr2+, and Cr3+ (Exception to Rules) In writing the electron configuration for Chromium the first two electrons will go in the 1s orbital. Therefore, one of the 4s2 electrons jumps to the 3d5 so that it is half-filled (see video below). The other school of thought is that the electron configuration of an element should be written so that the sublevels are in order of increasing energy, which will place the #"4s"# sublevel before the #"3d"# sublevel as indicated by the Aufbau diagram. Correct Electron Configuration for Chromium (Cr) Half-filled and fully filled subshell have got extra stability. ou bandido filmow Electron Configuration Exceptions - Chromium (Cr) & Copper. This has the advantage of writing the valence electrons (highest energy s and p sublevels) at the end of the electron configuration. WebThe electron configuration of the central chromium atom is described as. 
In these cases, a completely full or half full d sub-level is more stable than a partially filled d sub-level, so an electron from the 4s orbital is excited and rises to a 3d orbital. The ground state electronic configuration of the atom of an element. There are two main exceptions to electron configuration: chromium and copper. When written this way, all of the sublevels for #"n=3"# are written together in order of sublevels #3# s, p, and d, even though the #"4s"# sublevel fills before the #"3d"# sublevel according to the Aufbau principle. It is an exception to normal filling of orbitals in an atom. One is like the electron configuration of chromium in your question, in which the #"3d"# sublevel is written before the #"4s"# sublevel. When writing electron configurations using the Aufbau principle, there are two schools of thought. In atomic physics and quantum chemistry, electron configuration is the distribution of the electrons of an atom or molecule (or other physical structure) in. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
